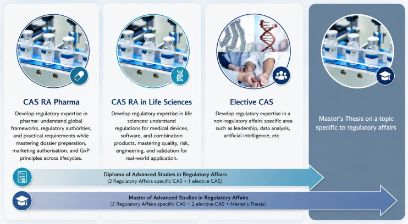
Content + Structure
This programme will provide you with the necessary expertise to work as a Regulatory Affairs Professional in the highly regulated environment of the medical technology, pharmaceutical and biotech industries.
Portrait
The healthcare industry evolved greatly over the last 10 years. As a result, the tasks of Regulatory Affairs Professionals have changed accordingly. New technologies such as artificial intelligence and machine learning (e.g., decision support systems), home virus detection kits, wearable patient monitoring devices, companion diagnostics and precision medicine are already being used today. In the wake of such innovations and the resulting revision or creation of new regulations, Regulatory Affairs Professionals must continually expand their knowledge.
To obtain and maintain regulatory approval of healthcare products, Regulatory Affairs Professionals must fulfill the following core tasks:
- Developing regulatory strategies
- Implementing regulatory measures
- Adhering to quality management
- Solving trade-related issues
- Negotiating/communicating with authorities
- Crisis management
Career prospects
With a Master of Advanced Studies in Regulatory Affairs, you will be qualified to take on a responsible position in the medical technology and pharmaceutical industry. The roles are not only critical for the success of companies – many of them are also legally required functions.
| Role | Sector | Main Responsibilities |
|---|---|---|
| Person Responsible for Regulatory Compliance (PRRC) | Medical Devices | Compliance with regulatory requirement, technical documentation, PMS/Vigilance obligation, QMS oversight |
| Regulatory Affairs Manager (RA) | Pharmaceuticals | Regulatory submissions, communications with authorities, lifecycle management of approvals |
| Qualified Person (QP) | Pharmaceuticals | Batch release, ensuring GMP compliance, responsibility for manufacturing conformity |
| Qualified Prson for Pharmacovigilance (QPPV) | Pharmaceuticals | Establishment and oversight of the PV system, evaluation of safety data, authority communication |
While these roles often require additional practical experience in the industries before you can take them on in full, the MAS Regulatory Affairs provides you with the essential regulatory knowledge and qualifications to grow into these roles step by step.
Statement

Christina Degen-Wendlinger
Senior Consultant, inmedis GmbH
“I am impressed how the students of the CAS Regulatory Affairs in Life Sciences combine the knowledge acquired in the seminars with practice in their case studies, resulting in well-founded new implementation solutions. In my opinion, this is what makes the MAS Regulatory Affairs stand out. The students are specifically prepared for the challenges of their working world.”