Structure + Content
The CAS Regulatory Affairs in Life Sciences focuses on the regulations for medical devices, in vitro diagnostics, and medical software.
Objectives + Skills
Environment
- Regulated environment in medical technology and the life sciences industry.
- Quality and risk management in medical technology and the life sciences industry.
Medical devices including software as a medical device, active pharmaceutical ingredients, and medicinal drug products as well as combination products (medical devices – drugs/biologics) are governed by diverse standards and regulations. This CAS specifically addresses the regulations for medical devices, in vitro diagnostics, and medical software.
For a comprehensive understanding of pharmaceutical product regulations, we recommend the CAS Regulatory Affairs Pharma.
Course overview
This CAS comprises a course block covering EU, CH, and global regulations. In addition, there is a specific course block on the US regulatory requirements for medical devices (FDA).
To enhance learning, participants carry out a semester project (Living Case) of a topic of their choice, preferably from their professional environment.
Both course blocks follow the phases of the regulatory life cycle of medical devices, in vitro diagnostics, and medical software. Essential regulations, standards, norms, and guidelines relevant to each phase are addressed during the specific teaching modules.
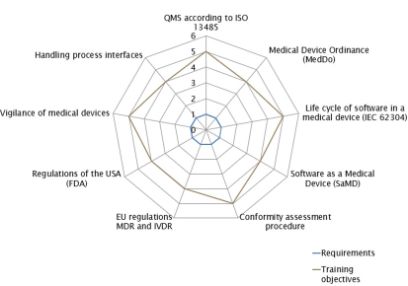
Training objectives
- You gain comprehensive knowledge of the EU Medical Device Regulation (MDR) and the EU In Vitro Diagnostic Medical Devices Regulation (IVDR) and demonstrate proficiency in their practical application.
- You acquire the necessary expertise to establish a quality management system (QMS) for medical devices, aligning with ISO 13485.
- You know the interfaces to processes such as risk management, usability engineering, design verification, design validation, process validations and reviews.
- You will develop the ability to implement national regulations on reporting obligations and corrective measures in the event of serious incidents involving medical devices.
Testimonial

The continuing education program in Regulatory Affairs in Pharma and Life Sciences provides an excellent foundation for understanding global regulatory frameworks. It is helping me strengthen my expertise in regulatory affairs and bridge my prior experience in pharmacovigilance with new regulatory knowledge. It offers a perfect balance of academic and real-world learning.